New catalyst can facilitate urea assisted water splitting – new strategy for energy efficient hydrogen production
Scientists have identified a new
catalyst that can efficiently oxidize urea and lower the energy demand for
hydrogen generation by urea-assisted water splitting thereby making way for
improved production of the green fuel.
Understanding
the importance of hydrogen energy in reversing climate change, the scientific
community is intensifying efforts to revolutionize hydrogen production, a key
player in the clean energy landscape. Electrolytic generation of hydrogen at
cathode, while inherently clean and green, has been hampered by the energy
demands of the oxygen evolution reaction at the anode (counter electrode).
A viable solution emerges from replacing the oxygen evolution reaction
with other anodic processes such as urea electro-oxidation reaction (UOR)
possessing lesser overall cell potential. By adding urea to water, it has
practically been shown to reduce the energy demand for electrochemical hydrogen
production by about 30%. This not only reduces the electrical energy input and
hence, the cost for hydrogen generation from water but also holds a promise for
remediating urea from wastewater in conjunction with energy generation while
converting urea into nitrogen, carbonate, and water. Despite the potential
advantages of this reaction, the catalysts developed so far not stable
vulnerable to COx poisons (by-products of UOR) posing barriers to industry-scale
implementation of this process.
A team of
scientists from Centre for Nano and Soft Matter Sciences (CeNS), Bengaluru – Nikhil
N Rao, Dr Alex Chandraraj and Dr Neena S John, have demonstrated a non-noble
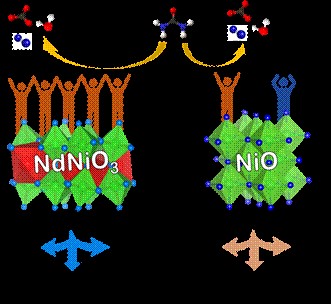
metal catalyst, Ni3+-rich
– Neodymium Nickelate (NdNiO3) with metallic conductivity that efficiently oxidizes
urea, thereby lowering the energy demand for hydrogen generation by urea-assisted
water splitting.
The
investigation was taken up as part of an ongoing project to develop high-active
and tolerant catalysts based on high-valent Ni-oxides for urea electrolysis,
which is supported by the erstwhile Science and Engineering Research Board
(SERB), now ANRF. The team used neodymium nickelate as an electrocatalyst for
UOR, and using techniques such as X-ray absorption spectroscopy,
electrochemical impedance spectroscopy, and Raman spectroscopy performed operando (under
operating conditions), substantiated that the catalyst drives the reaction
specifically through a ‘direct mechanism’. The direct mechanism exhibited by
electrochemically activated neodymium nickelate stands out for its minimal
catalyst degeneration and reconstruction, contrasting with the indirect
mechanism requiring regeneration after each cycle of UOR that prevails in Ni2+-rich catalysts such
as NiO. The catalyst has superior reaction kinetics (making the reaction
faster), and enhanced stability during prolonged electrolysis, which are the
attributes of a good electrocatalyst.
Towards
addressing the challenge posed by COx poisons, which are known for deactivating
UOR catalysts and compromising their long-term electrolysis durability,
neodymium nickelate emerges as a promising solution. Its exceptional tolerance
to COx poisons endows it with notable electrocatalytic stability. Computational
calculations in collaboration with Dr Moumita Mukherjee and Prof Ayan Datta
from Indian Association for the Cultivation of Science (IACS), Kolkata,
validate the experimental findings.
Published
in ACS Catalysis, a journal dedicated to publishing experimental and
theoretical research on catalytic materials, this work could direct future
studies aiming to enhance the number of NiOOH species and stabilize these
species on Ni3+-rich substrates. The goal is to achieve improved
performance with low mass loading of active Ni in the catalyst, marking a
significant step towards sustainable and efficient hydrogen production.
[Publication
link: https://doi.org/10.1021/acscatal.3c04967]